Leucine MES · Pharma Manufacturing · 2020 — Present
The OS of pharma manufacturing.
Six years, 51 customers, 370 plants.
I joined as Day-1 designer in 2020 and built it from Digital Work Instructions for 4 pharma customers to a full Manufacturing Execution System for 51 regulated enterprises across 4 continents.

Role
Head of Design
Timeline
2020 – Present (6+ years)
Team
5
Scope
Enterprise SaaS
Background
Day One. Sole designer.
A pharma product I knew nothing about.
I joined Leucine in 2020 with a single design seat. The product was DWI — Digital Work Instructions, paper-on-glass for shopfloor operators on industrial tablets. The wedge was 21 CFR Part 11 compliance plus a stripped-down prototyping module that let pharma teams compose their own SOPs. I had one obvious thing working against me — zero pharma background — and one thing working for me: the founder said yes to a strategy I argued for in week three.
Six years later, the same primitives I drew in 2020 run an entire MES platform: DWI → ePBR → eBMR → full Manufacturing Execution System → LeucineOS. One platform, three hubs (Manufacturing / Quality / Lab), one ontology underneath. I designed it from solo hire through Head of Design.
Impact
Six years on one platform. The primitives I drew in 2020 today run 51 pharma enterprises and 370+ GMP sites globally. Cipla alone has 8,000+ concurrent operators on the platform; Huvepharma audited a 92% reduction in uncontrolled data entry against their paper baseline; senior operators in 4 continents validate the same execution surface.
The mistake that taught the platform
From one object to two
checklist as template, job as instance.


In the first version, a checklist wasthe job. Within four months every customer plant had local SOP variants. Cipla Indore's tablet-coating cleaning was a different checklist from Cipla Goa's, even though the master procedure was identical. QA reviewers were drowning — the same SOP existed as 40 forks across 12 sites. The product was getting harder to use the more it was used.
The fix was to separate master (the template) from instance(the executed job). I rebuilt the data model and the UI in the same month. I didn't know it then, but ISA-88 had codified this exact separation in 1995. The lesson: if your system feels harder to use the more it's used, the abstraction is wrong, not the surface.
The alphabet
DWI → ePBR → eBMR → MES → LeucineOS.
Same primitives. New use cases.
Six years of acronym creep. Every step extended the platform without replacing it. The same task object, signature object, and audit trail that ran a cleaning checklist in 2020 today runs a 14-stage tablet manufacturing batch at Cipla. Compounding, not rebuilding.
2020 · Year 1
DWI — Digital Work Instructions
Paper-on-glass. Operators executing SOPs on industrial tablets, 21 CFR Part 11 compliant. Four customers, $6,000 per facility. The primitives — task, signature, audit trail, mandatory block — were set here.
2022
ePBR — Electronic Production Batch Record
First module bridge. Cleaning checklists rolled up into the production batch. I extended the master/instance pattern from a single SOP to a hierarchy — procedure → unit operation → phase. Same primitives, deeper tree.
2023
eBMR — Electronic Batch Manufacturing Record
Full batch record digitization. Stage chaining, weighing-balance integration, equipment usage logs, regulator-signable PDF. I added structured tolerance, IPC primitives, and dual signature to the same execution surface.
2024
MES — Manufacturing Execution System
The product crossed the line from digital paperwork to execution layer. Equipment integration, IoT andon lights, real-time job orchestration, multi-site coordination. The wedge had eaten the category.
2025–present
LeucineOS — Manufacturing + Quality + Lab Hubs
Three hubs unified: Manufacturing (MES), Quality (CLEEN, deviations, change control), Lab (LIMS-adjacent execution). One ontology underneath. One Intelligence Hub on top.
June 2025 · The role bifurcated
From designing every screen
to designing the world model agents reason against.
Before June 2025
I designed every screen in every module. The team executed; I led design.
After June 2025
I design the world model the AI agents reason against. The team owns the screens; I own the ontology and the AI surface.
The work
What 6 years of compounding produced.
Mature execution surface today. Below: the master record being designed (1), the operator execution surface evolving from Year-1 to 2026 (2–3), the integrated logbook in live view and builder mode (4), the plant-manager dashboard (5), and the process interlock that hard-blocks an out-of-spec batch with a Level-3 QA override path (6). Same primitives as 2020, recomposed across nine modules.

Before — 2020

After — 2026

Logbook — live view

Logbook — builder



Design decisions
Ten decisions the platform compounded on.
Not the only ten — the ten the system bent around. Each one started as a customer escalation, an audit finding, or a flat refusal from a plant manager. Each one became a primitive every later module reused.
Decision
What it reveals
Master / instance separation
Rebuilt ISA-88 from first principles before knowing the standard existed. The bug taught the architecture.
Industrial-tablet-first interaction model
Hardware constraints come first, aesthetics second. When operators route around the product back to paper, the form factor lost.
Mandatory-block hard refusal
Forcing the operator to face the gap at task time is cheaper than forcing QA to face it at batch-release time.
Stage hierarchy as the unit of execution
The hierarchy is the audit trail. Reviewers think in stages; the product had to mirror that or lose them at review.
Prototyping module — no-code SOP builder
The differentiator that closed the first four customers. Without it Leucine ships bespoke checklists; with it, the platform is buyable.
Pre-assembled logbook templates
Cut onboarding from weeks to days. Removed the single biggest reason new customers churned in month one.
Camera OCR on weighing balances
Any analog readout can become structured input without retrofitting hardware. Kill manual transcription, not the device.
Process Interlocks — declarative refusal
The system says no before the operator can. Cleaner than training, cheaper than auditing, fires every time at 3am.
Inbox — unified QA review queue
QA is the bottleneck of every batch release. Concentrating their work in one queue compressed review cycles ~40% at Cipla.
Ontology-first data model
Every new module shipped faster — pattern reuse, shared entities, no module-by-module data forks. Built defensively in 2022; turned out to be the substrate Cortex AI agents later reason against.
What I argued for and lost
I pitched a full prototype review flow with branching approval and inline annotation. Founder cut it at deal-close to compress scope. The stripped 3-pane builder shipped instead and closed Zydus + 3 more customers in Year 1. The cut was right; the argument made the design defensible. Full chapter in Deep Dive.
Context
Validated by senior operators
across four continents.
Cipla
Dr. Reddy's
Amneal
Biocon
Valent BioSciences
Revlon
Viatris
Zydus
Lupin
Shopfloor reality
What the platform looks like in production — gowned operators on the wall screen reviewing a live Packaging Batch Record on Leucine, mid-batch.
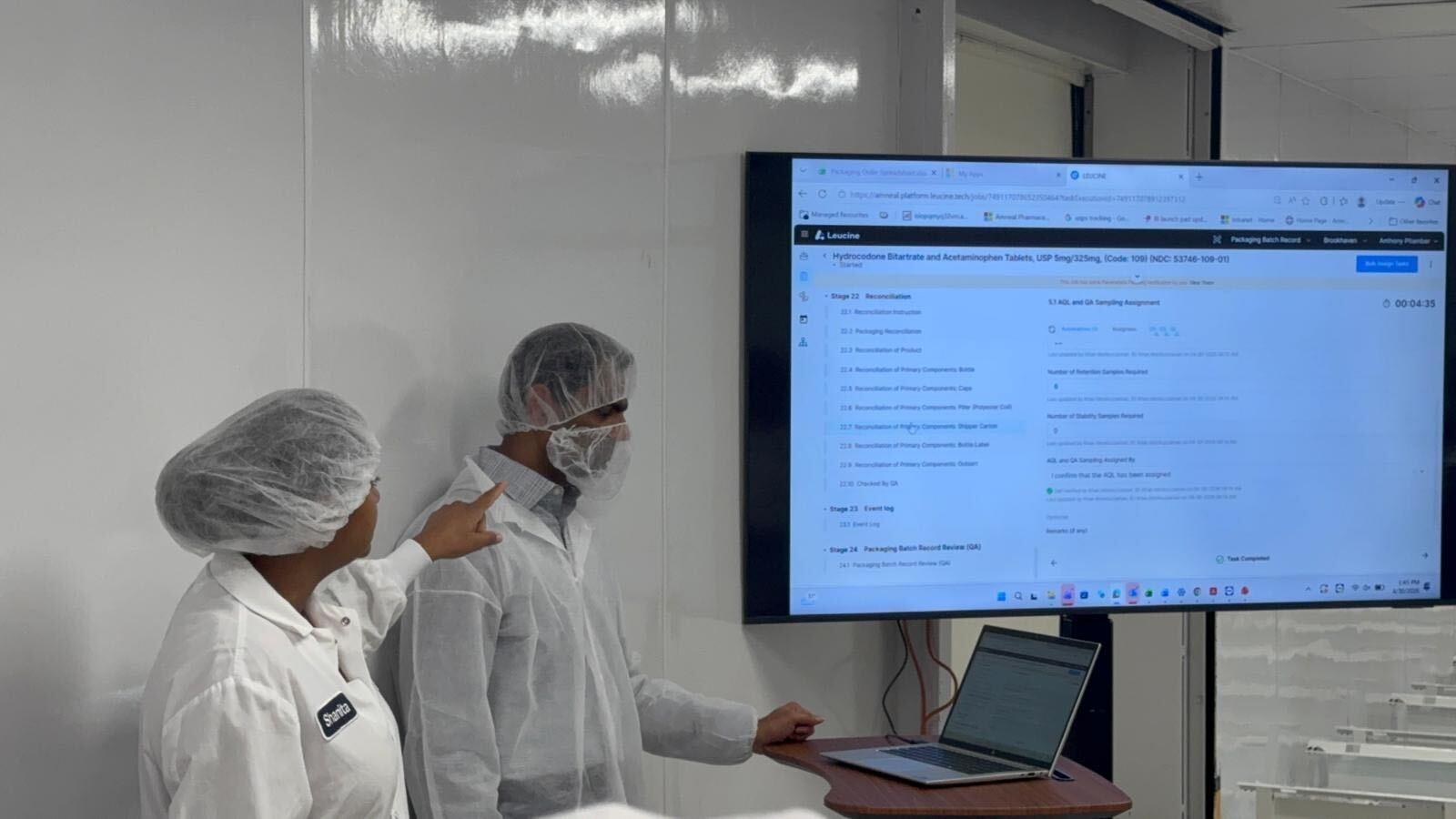
Operators at the wall-mounted screen, reviewing a packaging batch record stage live.

Packaging corridor — the surface I designed in 2020 in front of the people who actually use it.
Commercial frame
Most pharma MES rollouts take 18–36 months and 30–60% fail to reach planned scope. Leucine landed on the operator surface first, then extended down to the substrate.
| Vendor | Implementation | Risk profile |
|---|---|---|
| Körber PAS-X (Werum) | 18–36 mo | 30–60% scope failure |
| Rockwell PharmaSuite | 18–30 mo | Heavy IT integration debt |
| Tulip POG | 3–6 mo | Light footprint, GMP coverage gaps |
| Leucine MES | 2–6 mo | Newer category entrant |
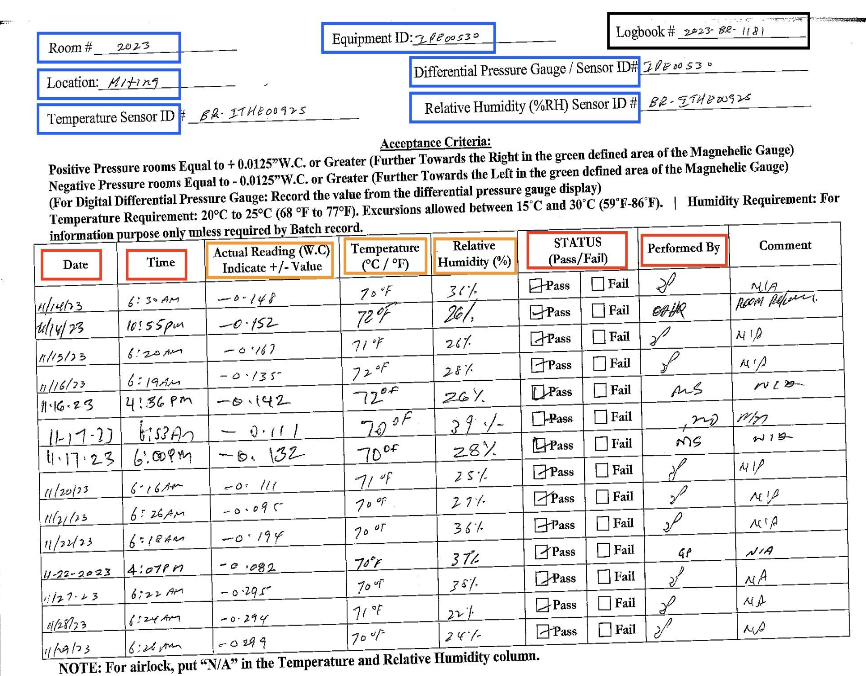
Operator empathy — Valent BioSciences, Iowa
“Our operators love it. They actually want the digital tablet on their station now. That wasn't true with the last system.”
Plant Manager — Iowa cleanroom rollout
Competitive benchmark — Sopharma, Bulgaria
“Leucine moves faster than the four MES vendors we evaluated. The architecture is cleaner. The operators reach proficiency quicker.”
Ed Bell, Senior Ops — post-evaluation against Werum, Rockwell, Siemens, Emerson

92%
Data Entry Reduction
100%
Global Harmonization
Manual fields
Eliminated
Automated
Controlled
Huvepharma audit. 15 manually-transcribed paper fields collapse into 1 controlled subtotal — 92% reduction in uncontrolled data entry, 100% global harmonization across plants.
Closing
This is what one designer can do when given six years inside one rigid domain — and the right co-conspirators.
Deep dive
The 6-year story,
for readers who want it.
Eight chapters. Top to bottom. No clicks.
Chapter 1 · Cipla Indore
The shopfloor visit
that reset four years.

Note — I couldn't take photos of the actual production floor; phones aren't permitted inside the GMP facility. I snuck a phone in and grabbed this in a corridor where it was just me, our PM, and a junior product designer from my team who was working on a feature.
Operator, Cipla Indore — February 2022
We still write everything on paper because if the tablet crashes mid-batch the digital record is gone, and at audit time it's only the paper a regulator will accept as proof.
That sentence reset every assumption from week one. The next four years were spent making the digital record more trustworthy than the paper one — not just compliant. Crash recovery, dual persistence, supervisor exception flow, signature attestation, structured deviation logging. All of it traces back to one operator, one tablet, one piece of tape.
Chapter 2 · The killed feature
What I argued the founder out of
and what shipped instead.
The pitch — full prototype review flow

What shipped — stripped-down builder

What it taught me
The founder was right to compress. Scope discipline at deal-close beats feature richness at demo. The argument made the design defensible — but the cut shipped the company.
I pushed for the full review flow. The founder pushed back on scope. We landed on the stripped-down builder — three panes, drag a primitive, configure, save. It closed Zydus, then three more customers in Year 1. The full review flow eventually shipped in Year 3, when the platform could carry the weight.
Chapter 3 · Team arc
From one seat
to a team to a systems architect.
2020 – 2022
One designer
Day-1 hire. Sole design seat. Shipped DWI to four customers. Argued the prototyping module wedge with the founder. First Cipla shopfloor visit.
2023 – 2025
Five-person team
Hired and grew the team to five. Onboarded the design system from notes-in-Figma to a shared component library. Critique cadence, design QA, research function. Shipped ePBR, eBMR, early MES.
2025 – present
Lead + Systems Architect
Cortex shipped. Role bifurcated. Lead Designer (managing the team that ships modules) + Systems Architect (designing the ontology and the AI surface). Team owns the screens; I own the world model.
The team scaled in step with the platform — not faster. Three years solo gave the architecture time to harden before more designers could safely extend it. Hiring earlier would have duplicated the master/instance debt across more pairs of hands.
Chapter 4 · The ontology bet
Why I built the substrate
before anyone asked for it.
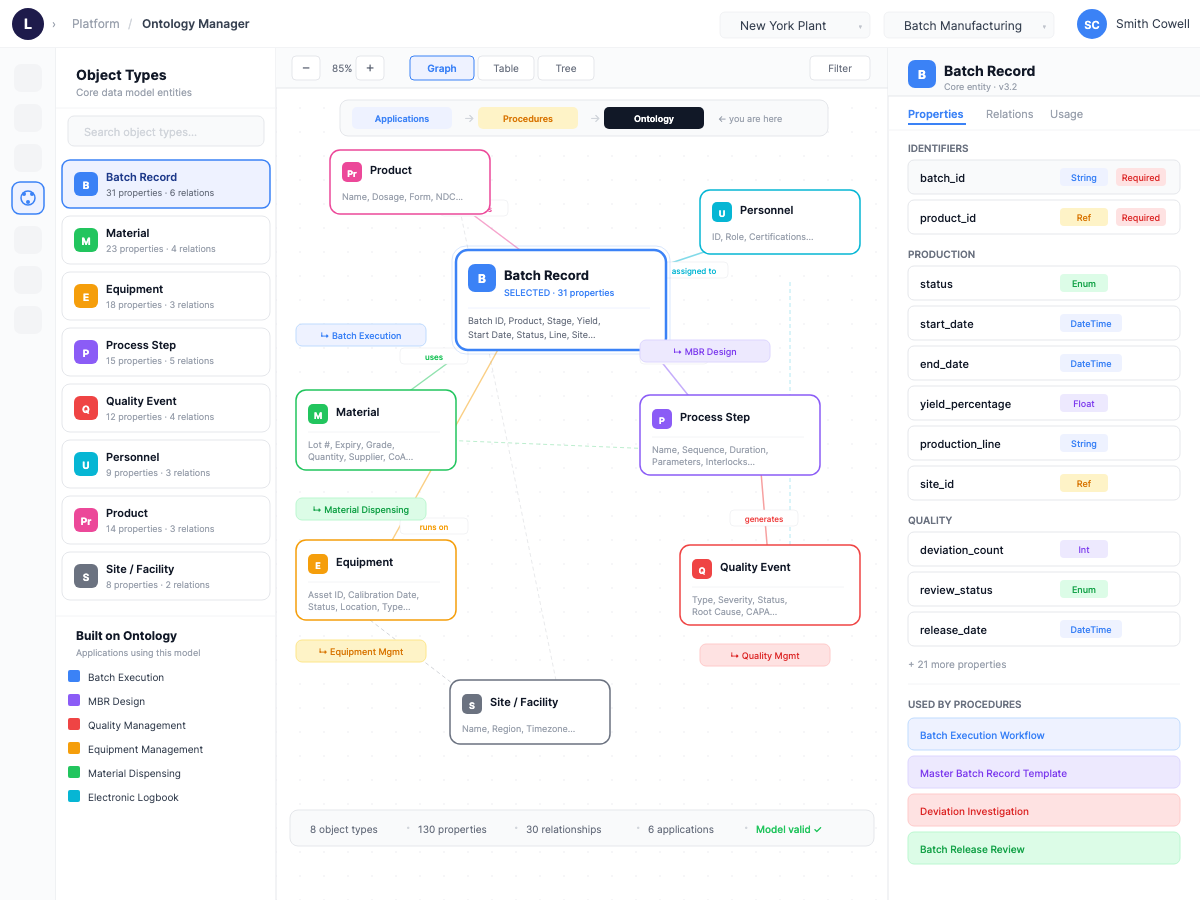
Ontology Manager — object graph
Connected Logbooks — substrate in motion

Two views of the same substrate. Left — the ontology defined: universal entities (Batch Record, Material, Equipment, Process Step, Quality Event) that every module references instead of forking its own data model. Right — the ontology in motion: live sync across MES, QMS, and ERP keeps every entity coherent across systems. Together they prove the substrate isn't theoretical. It's the running platform.
What the ontology is
Universal entities — Equipment, Material, Procedure, Batch, Personnel, Site — referenced by every module instead of forked per module.
What it unlocked
30% smaller code per new module. Cross-module queries in one statement. The substrate Cortex AI agents reason against.
I built the ontology defensively in 2022 to stop modules duplicating themselves. In 2025 it turned out to be the exact thing AI agents needed to reason against. The bets that compound aren't always the ones you can pitch at the time.
Chapter 5 · The June 2025 inflection
AI changed what the design surface had to be.

Anthropic released Claude 4 capable of reliably operating structured tools. Two layers met — the ontology I'd built defensively, and the agents that needed a structured world to reason against. The role bifurcated inside a quarter (visualised up in The alphabet section). Cortex itself was built by a separate engineering team; what I designed is the world model the agents reason against and the surface where humans approve their work.
Chapter 6 · The ISA-88 fix I rebuilt by accident
Rebuilding a 25-year-old international standard
from first principles.

The lesson
If your system feels harder to use the more it's used, the abstraction is wrong, not the surface.
ISA-88 was published in 1995. It mandates a four-tier hierarchy (procedure → unit procedure → operation → phase) and strict separation between procedural model (template) and equipment model (instance). I didn't know it existed when I shipped DWI v1. The bug forced the same separation. Reading the standard on day one would have compressed three years into eighteen months.
Chapter 7 · What an MES actually is
The category, the entrenched vendors,
and why most MES rollouts fail.
| Vendor | Founded | Implementation | Risk profile |
|---|---|---|---|
| Körber PAS-X (Werum) | 1969 | 18–36 mo | 30–60% scope failure |
| Rockwell PharmaSuite | 2004 | 18–30 mo | Heavy IT integration debt |
| Siemens Opcenter | 2018 (was Camstar) | 18–24 mo | Configuration vs customisation gap |
| Emerson Syncade | 2008 | 12–24 mo | Strong DCS, weak operator UX |
| SAP ME / MII | 2007 | 24–36 mo | ERP-shaped, not plant-shaped |
| Tulip POG | 2014 | 3–6 mo | Light footprint, GMP coverage gaps |
| Leucine MES | 2020 | 2–6 mo | Newer category entrant |
Manufacturing Execution Systems sit between ERP (what to make) and the shopfloor (the doing). 30–60% of pharma MES implementations fail to reach planned scope. The reasons are consistent: misaligned ISA-88 modelling, change-control debt, weighing-balance and PLC integration overhead, and the gap between the system IT buys and the system the operator uses. Leucine's wedge was the operator surface. The ontology was the structural answer.
Chapter 8 · What I'd do differently
Three lessons I'd hand to anyone
starting a regulated-domain platform tomorrow.
01
Read the standards earlier
ISA-88, ISA-95, GAMP 5 Cat 4, ALCOA+. I rebuilt half of them by accident. Reading them on day one would have compressed three years into eighteen months.
02
Visit the shopfloor in week two, not month eighteen
The Cipla Indore visit reset four years. Doing it earlier would have saved a year of designs that didn't survive contact with gloved operators.
03
Treat the ontology as architecture, not as a refactor
Year-4 me retroactively unified data models that should have been one model from the start. The substrate is the architecture; build it first.
The substrate is the architecture. Build it first, read the standards, walk the floor in week two. Three lessons I now reflexively apply on any new regulated-domain project — and three lessons I'd pay anyone who's about to start a similar build to skip past.